Key Points
- Long-acting HIV medicines have become increasingly popular in recent years, offering a safe way to reduce how often someone needs to take HIV medicines.
- Long-acting HIV medicines are injected by a healthcare provider rather than taken as an oral pill at home.
- Unlike most oral HIV medicines that are taken daily, long-acting HIV medicines are injected every two weeks to every six months, depending on the medicine.
What are long-acting HIV medicines?
Long-acting HIV medicines are HIV medicines that stay in the body longer than traditional HIV medicines. Because they last longer, they can be taken less frequently.
Long-acting HIV medicines include:
As of April 2026, these are the only long-acting medicines that are approved by the U.S. Food and Drug Administration (FDA) for HIV prevention or treatment. This fact sheet focuses on HIV treatment. View the HIVinfo Pre-Exposure Prophylaxis fact sheet to learn more about long-acting HIV prevention methods.
How are long-acting HIV medicines different?
Long-acting HIV medicines are different because they stay in the body and keep working for much longer than pills that need to be taken every day. Some long-acting treatment plans can reduce or even replace the need for daily pills, and others can be used along with everyday pills.
Instead of taking pills every day, long-acting HIV medicines are given as injections much less frequently. The current FDA-approved options are usually given on the following schedule:
- Cabenuva – Every one or two months
- Sunlenca – Every six months
- Trogarzo – Every two weeks
Unlike HIV medicines in pill form, long-acting HIV medicines are given by a healthcare provider at regular appointments. Research suggests that long-acting HIV medicines may help some people with taking their medicine as prescribed and on schedule. This is referred to as treatment adherence.
Treatment adherence is important for HIV medicines to work as well as possible. However, taking a pill every day does not work for everyone. Long-acting HIV medicines may help some people overcome barriers to daily treatment adherence, such as busy or unpredictable schedules that can make it difficult to take medicine every day.
Scientists continue to study and monitor new long-acting medicines for HIV prevention and treatment to better understand how they may benefit different people, including those who prefer an option other than a daily pill. The HIV Treatment Adherence fact sheet provides more information on factors that may affect adherence.
Are long-acting HIV medicines safe?
Long-acting HIV medicines, like all other FDA-approved medicines, are considered safe and effective for most people. Before getting injections, most long-acting HIV medicines must be taken as pills for a short time (called an oral lead-in) to ensure the medicine is well-tolerated.
However, all medicines have the potential for some side effects. For long-acting HIV medicines, some common side effects include:
- Diarrhea
- Dizziness
- Fatigue
- Fever
- Headache
- Nausea
- Rash
- Sleep problems
- Soreness, swelling, or redness around the injection site
Less commonly, long-acting HIV medicines may cause more serious side effects like depression or liver problems. However, serious side effects are rare and can usually be managed with help from your healthcare provider.
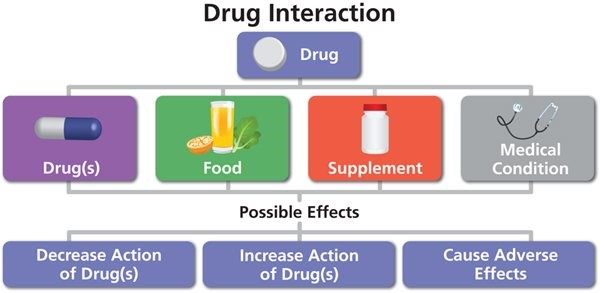
In addition, many HIV medicines (including long-acting HIV medicines) may be prone to drug interactions if combined with other medicines, including over-the-counter medicines. For example, long-acting HIV medicines may interact with some antibiotics and anticonvulsants. See the HIVinfo fact sheet What is a Drug Interaction? for more information on drug interactions and HIV medicines.
Because there are generally not enough data to know whether these medicines are safe during pregnancy or while breastfeeding, they are often not recommended in these situations. If you are pregnant, planning a pregnancy, or breastfeeding, be sure to talk to your healthcare provider about the safest treatment options for you before starting any new medicines.
Should I switch to a long-acting HIV medicine?
Switching HIV medicines should occur only after consultation with a healthcare provider. If a daily HIV treatment regimen is working effectively and is tolerable, switching may not be necessary. Switching HIV treatment regimens may lead to drug resistance, which could limit treatment options in the future.
In addition, there are only a few long-acting HIV medicines available to choose from, with some options reserved for unique situations. For example, Trogarzo is typically prescribed when a person is resistant to multiple classes of HIV medicines. When this happens, commonly used HIV medicines do not work effectively, and Trogarzo is used as the next available option.
If you believe you are a good candidate for long-acting HIV medicine, talk to your healthcare provider to explore your options. Make sure to discuss your circumstances, lifestyle, preferences, and your full medical history so your provider can get a clear sense of what would work best for you.
Always consult your healthcare provider about taking any new medicines and any concerns related to an HIV treatment regimen.
This fact sheet is based on information from the following sources:
From the National Institutes of Health:
From CDC:
From the World Health Organization:
Also see the HIV Source collection of HIV information and resources.